IBC FAQs
Getting Started
When do I need IBC oversight? What are IBC materials?
IBC approval needs to be obtained prior to using IBC materials.
IBC materials are defined as:
- recombinant or synthetic nucleic acids
- organisms or viruses containing recombinant or synthetic nucleic acids
- materials that are biohazardous (i.e., those that are both hazardous to a human being and that are biological or come from a biological source)
The following is a non-exhaustive list of IBC materials. If you have a question whether something is an IBC material, please contact an IBC Administrator to ask. This is your most accurate and effective route to an answer.
- Use of recombinant or synthetic nucleic acids (e.g., plasmids, bacterial artificial chromosomes, etc.) in bacteria, eukaryotic cells, plants, animals, or other forms of life
- Use of recombinant or synthetic viral vectors (e.g., AAV, lentivirus, retroviruses, etc.)
- Use of gene drive techniques or technologies
- Use of animal cell lines, tissues, or animals that either transiently or stably include recombinant or synthetic nucleic acid materials
- Transgenic animals (whether stably or transiently transformed)
- Animals that are hosting transgenic cell lines or microorganisms, carrying biological or technological conveyances that contain transgenic cells or microorganisms, etc.
- Non-transgenic animals being used with transgenic bacteria, viruses, or other biological agents or with biological toxins
- Transgenic plants (whether stably or transiently transformed)
- Human gene therapy
- Research involving pathogenic or non-pathogenic microorganisms
- Use of samples that may contain pathogenic microorganisms (e.g., soil, water, bodily fluids, etc.)
- Use of human or non-human primate samples, cell lines, tissues, or fluids
- Use of biological toxins
I’m new to Rice or I plan to start working with recombinant or synthetic nucleic acids or biohazardous materials for the first time. Where do I get started?
If you plan on starting work on IBC materials at Rice University, the best thing to do is to contact an IBC Administrator o discuss your research. You will also need to meet with the Environmental Health Safety and Laboratory Operations team to ensure that the facilities are appropriate and that the personnel have sufficient training.
The following infographic shows the general process for onboarding PIs who have not previously had IBC oversight of their research. It provides a simple visual method for your understanding.
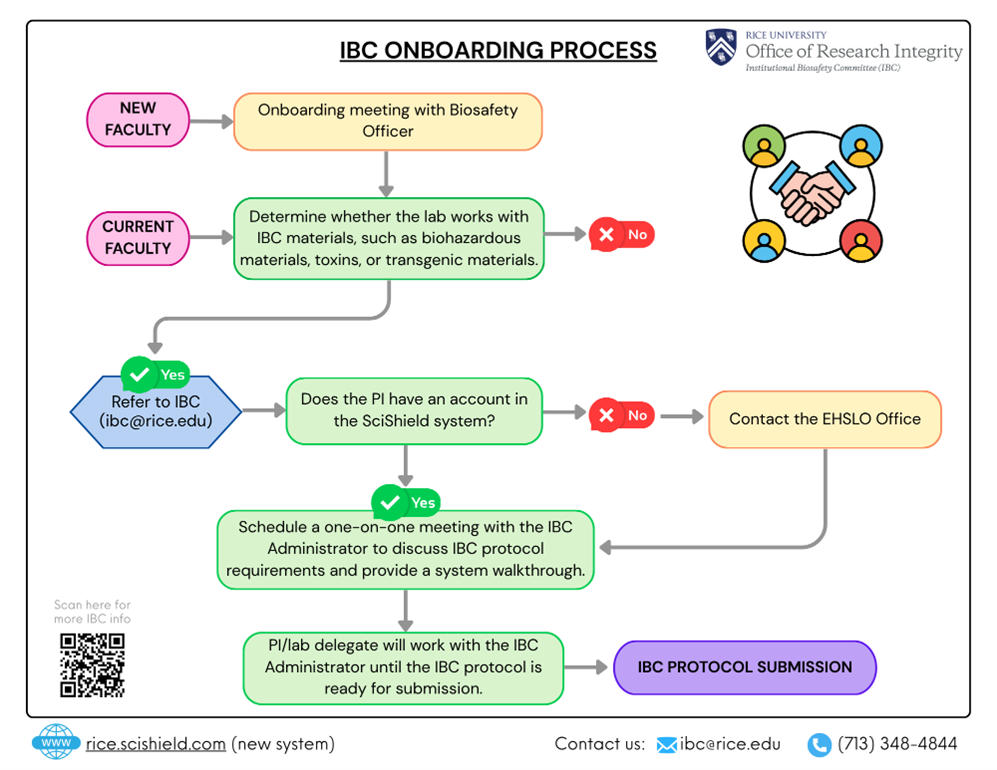
How do I submit an IBC protocol?
IBC protocols are submitted on the SciShield website. If you need assistance in navigating the system or guidance on what information to provide, please see below or contact an IBC Administrator.
How do I submit a Modification to my IBC protocol?
If you find that your IBC protocol does not describe research that you would like to do (i.e., materials, processes, or personnel are absent), you should file an IBC Protocol Modification to request permission to perform the necessary research.
Modifications to IBC protocols are straightforward and common and, in many cases, can be approved quickly. Additionally, Modifications are necessary to ensure that your research remains compliant with Rice policies and NIH guidelines.
Instructions for filing an IBC Protocol Modification can be found by clicking here. If you have questions about whether a Modification is necessary or how to file a Modification, please contact an IBC Administrator.
What should be in my IBC protocol?
What kinds of information needs to be included in an IBC protocol?
Your IBC protocol should include sufficient information for the IBC to understand and evaluate the risks associated with the research that you and your researchers will be doing. This means that the IBC needs to know who will be doing what type of experiments with which IBC materials and where the experiments will be performed.
To this end, the following information is required in an IBC protocol.
- Objectives: Describe purposes/goals of the project.
- Planned Experiments: Provide a summary of the experiments you intend to conduct.
- Research Location: Specify the location(s) where the research will be conducted.
- Research Team: Specify the people who will be doing the research.
- Appendix A: Describes all of the recombinant, synthetic, or transgenic materials being used in the research, as required by Section III-D of the NIH Guidelines.
- Appendix B: Describes all of the biohazardous materials being used in the research. (If no biohazardous materials are being used, an Appendix B form is not required.)
- Viral Vector Form: Describes the nature of the viral vectors being used in the research. (If no viral vectors are being used, this form is not required.)
- Risk Assessment: Describes safety measures and practices used to mitigate the potential hazards of the materials that are being used.
What information goes in my Appendix A?
Your Appendix A describes all of the recombinant, synthetic, or transgenic materials being used in the research, as required by Section III-D of the NIH Guidelines. This includes: the host of the material (e.g., bacteria, human cells, animals, etc.), the Risk Group for the host, the vector used to introduce the material into the host (e.g., plasmid, virus, etc.), the nucleic acid material targeted for introduction into the host, the source of the nucleic acid material (and an indication if the material is synthesized or cloned), the Risk Group of the source of this material, the intended purpose for putting this material into the host (e.g., expression, protein purification, gene silencing, protein visualization, etc.), the location where the material is being used and/or stored, and, if appropriate, a notation that the material is being stored but not actively used.
How can I check risk groups of the organisms I plan to work with?
Risk Groups for the materials that you are working with can be most authoritatively found in the NIH Guidelines in Appendix B. Please note that this list is not completely exhaustive; if you are not able to find the agent in question on this list, it does not mean that the organism is RG1. If you cannot find a determination, please contact an IBC Administrator.
What is the difference between Appendix A and Appendix B?
The Appendix A form is used to communicate the recombinant, synthetic, or transgenic materials being used in the research, as required by Section III-D of the NIH Guidelines. Appendix B is used to communicate the nature of the biohazardous materials that are used by the research group.
For example, a non-hazardous strain of E. coli containing GFP, it would be listed on Appendix A, but not on Appendix B. A non-transgenic strain of P. aeruginosa would be listed on Appendix B, but not on Appendix A. But a transgenic strain of S. aureus would be listed on both Appendices.
What is Appendix C?
Unlike Appendices A and B, the IBC does not have an Appendix C form. References to Appendix C, in the context of IBC review, refer to Appendix C of the NIH Guidelines, which discusses organisms that are Exempt from NIH Guidelines.
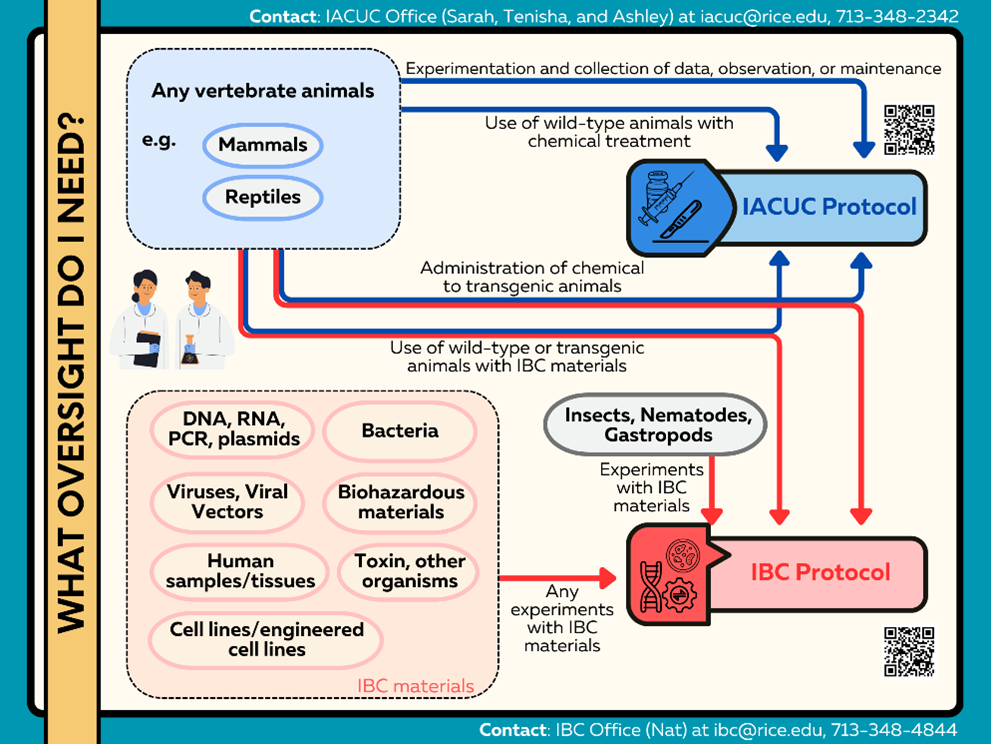
My experiments are related to animals, biohazard materials, and recombinant DNA. Which information should I include in the IBC protocol or the Institutional Animal Care and Use Committee (IACUC), or both?
Review the flowchart outlining which types of experiments require IBC approval, IACUC approval, or both. If you have any questions, please contact the IBC Office at ibc@rice.edu or the IACUC Office at iacuc@rice.edu.
PI Responsibilities and Requirements
Who can be a Principal Investigator of an IBC protocol?
A tenured, tenure-track, emeritus, or research faculty member with assigned research space at Rice who is responsible for overseeing the research conducted in their laboratory.
What are PI's responsibilities with IBC protocol based on the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules
PIs are responsible for ensuring full compliance with the NIH Guidelines during any research involving recombinant or synthetic nucleic acid molecules.
These requirements include:
- Be adequately trained in good microbiological techniques.
- Adhere to the IBC-approved research protocol and emergency plans for handling accidental spills and personnel contamination.
- Not initiating or modifying any recombinant or synthetic nucleic acid molecule research that requires IBC approval until the research or the proposed modification has been approved by the IBC and has met all other requirements of the NIH Guidelines.
- Comply with shipping requirements for recombinant or synthetic nucleic acid molecules.
- Report any significant problems, violations of the NIH Guidelines, or any significant research-related accidents and/or illnesses to the Biological Safety Officer, Greenhouse/Animal Facility Director (where applicable), and an IBC Administrator as soon as possible.
- Report any new information bearing on the NIH Guidelines to an IBC Administrator.
Additional information can be found in the NIH brochure.
Where can I find information on biosafety practices for each biosafety level and for various types of containment (plant, animal, and general)?
Containment Information
Standard BSL-1 containment practices can be found in the Biosafety in Microbiological and Biomedical Laboratories (BMBL) on pgs 32-36 and in The NIH Guidelines on pgs 76 and 77.
Standard BSL-2 containment practices can be found in the Biosafety in Microbiological and Biomedical Laboratories (BMBL) on pgs 37-43 and in The NIH Guidelines on pgs 77-79.
Standard ABSL-1 containment practices for use with ANIMALS can be found in the Biosafety in Microbiological and Biomedical Laboratories (BMBL) on pgs 71-78 and in The NIH Guidelines on pgs 119 and 120.
Standard ABSL-2 containment practices for use with ANIMALS can be found in the Biosafety in Microbiological and Biomedical Laboratories (BMBL) on pgs 78-87 and in The NIH Guidelines on pgs 120-122.
Standard BSL-1P containment practices for use with PLANTS can be found in The NIH Guidelines on pgs 107 and 108.
Standard BSL-2P containment practices for use with PLANTS can be found in The NIH Guidelines on pgs 108-110.
What training is required for performing research with IBC materials?
All personnel performing research with IBC materials need to complete 2 training courses (General Laboratory Safety and Biosafety and Bloodborne Pathogens) that are offered by the Office of Environmental Health & Safety and Laboratory Operations. Training in each course must be renewed on an annual basis.
Each person must complete each course in person for the initial training, but annual renewal can be completed online on the SciShield website.
Please note! Although PIs may not be working with IBC materials on a daily basis, they are required to maintain current training in these matters. Without appropriate training, it is impossible for a PI to ensure that their students are performing their research safely.
As a PI, what do I need to do if I am leaving Rice?
As soon as you know that you will be departing Rice, please complete the PI Departure Questionnaire. Also, contact the IBC Administrator to schedule a meeting to discuss disposition of all IBC materials (whether this involves transfer to another lab or appropriate disposal).
Please also review materials available from our partners at EHLSO for lab closeouts available at the EHSLO website.
Information about IBC Meeting Practices
When does the IBC meet? What are the deadlines for protocol submission?
IBC meeting dates and submission deadlines are listed on our website . Meetings are typically held via Zoom and are open to the public. Requests for the meeting link can be made by emailing an IBC Administrator at least two weeks prior to the meeting.
The deadline for submitting materials for consideration by the IBC is typically two weeks prior to the date of the meeting. (E.g., if the meeting will take place on December 10th, the submission deadline would be November 26th.)
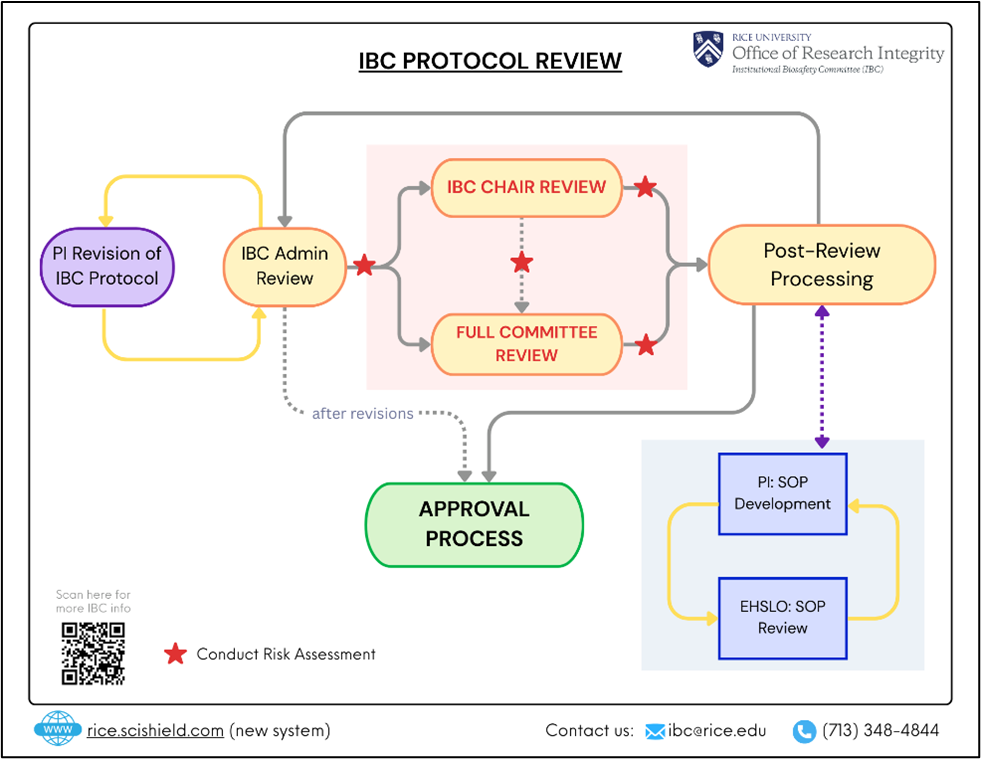
What is the next step in my protocol submission process after it has been reviewed by the committee?
This depends on the decision provided by the IBC. Four outcomes are possible from Committee review of an IBC protocol:
- Approval: When your protocol is Approved, you may begin your research, as described in the protocol, immediately.
- Approval Pending Revisions: When you receive this decision, there are minor problems with your IBC protocol that need to be addressed. The IBC will provide a letter describing the changes that are necessary. Changes should be completed within three weeks of receiving the decision letter, or your protocol application may be considered abandoned.
- Tabled: When the IBC tables a protocol, it generally means that there is insufficient information provided for the IBC to properly evaluate the risk and set containment criteria. In such a case, the protocol will be returned to the PI for revision. Please contact an IBC Administrator if you have questions about the changes required.
- Denied: This status represents the rare case where the IBC has determined that there is no safe way for the described research to be performed at Rice University. You may contact an IBC Administrator if you feel that this decision was made in error.
The following flowchart also provides a graphical summary of the review pathway for IBC protocols.
Acquiring Materials from Outside Rice
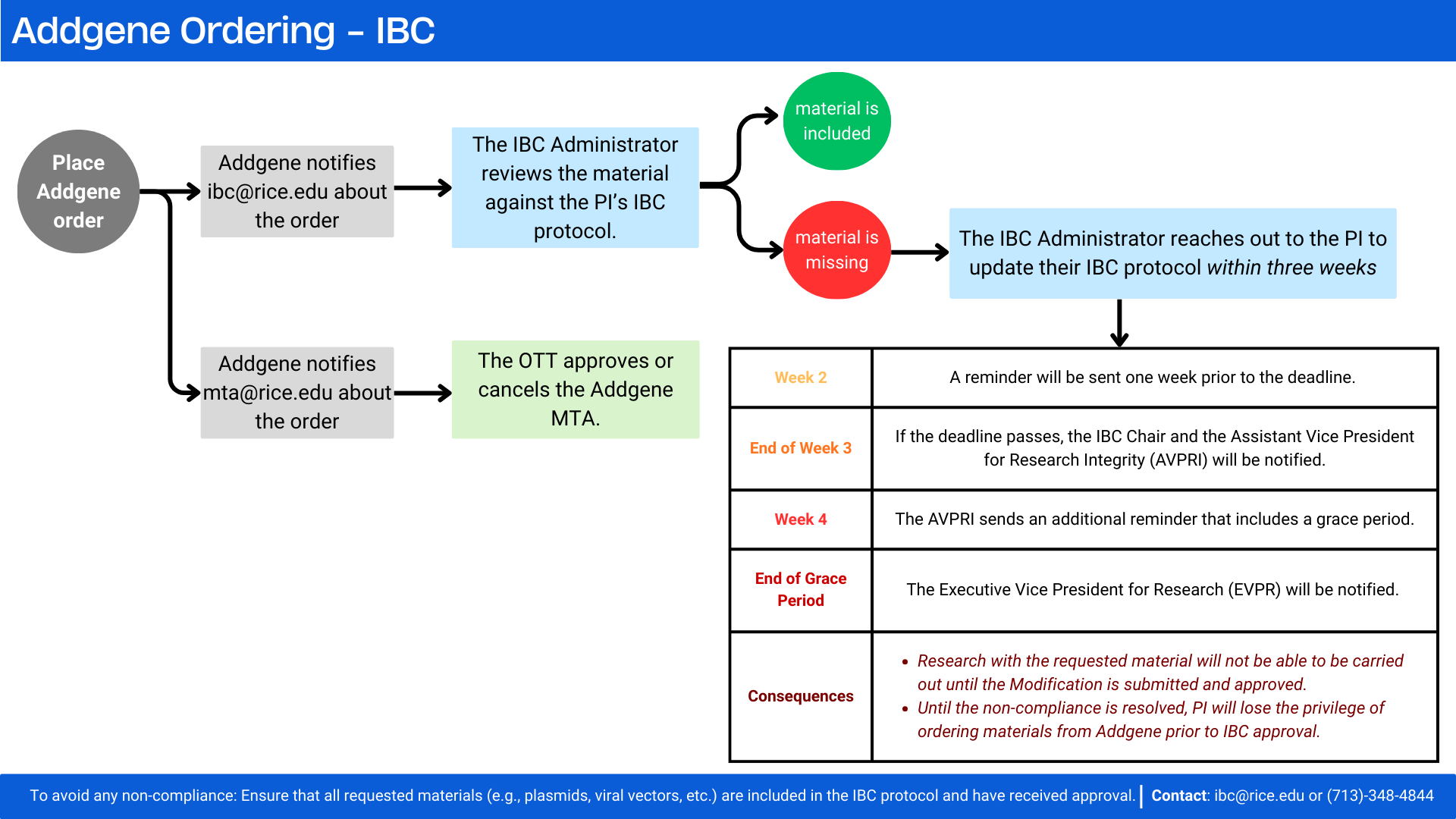
What do I need to do when I need to order plasmids from Addgene?
As with all materials, the materials being received from Addgene should be on your IBC protocol prior to ordering. If necessary, file an IBC protocol modification to receive approval for using the materials. (Instructions for this process can be found by clicking here.) You should have approval to use any materials prior to requesting them.
The IBC Office now reviews Addgene orders in the background when an order is placed. If the materials are not listed in the PI’s IBC protocol, the IBC Administrator will contact the PI to update the protocol.
We also provide a flowchart below for PIs that provides additional information for ordering materials from Addgene or other similar resources.
What steps should I take when receiving IBC materials from a collaborator?
As with all materials, the materials being received from a collaborator should be on your IBC protocol prior to receipt. If necessary, file an IBC protocol modification to receive approval for using the materials. (Instructions for this process can be found by clicking here.) You should have approval to use any materials prior to requesting them.
Please follow appropriate material transfer processes and complete any necessary material transfer agreements (MTAs). The Office of Technology Transfer is happy to assist with this process.
Need Help?
IBC Administrators are your first point of contact. They can help determine review requirements, walk you through submission, and escalate issues to the IBC Chair if needed.
📧 Email: ibc@rice.edu
📞 Phone: 713-348-4844
